#WeDeserveBetter recap: up to March 2023
- Narrative Review Shows Regulatory Inadequacies : Our analysis has revealed that the current CE marking fails to confirm if CGM systems are backed by sufficient clinical data for their intended uses. This was highlighted at ATTD 2023 and the presentation can be viewed here.
- Questionable CE Marking for Pediatric Use: Devices such as Glucomen Day, Medtrum A6, and GlucoRx Aidex have CE marks for paediatric use despite lacking published clinical accuracy data. We have formally requested MHRA to investigate.
- Lack of International Study Design Standards: Currently, there is no globally accepted standard for study design. This loophole allows devices intended for Type 1 Diabetes to be approved based on studies predominantly involving Type 2 patients with no meal or insulin challenges.
- Performance Standards Are Absent: The absence of internationally accepted performance standards has led regulators to grant non-adjunctive indications (no confirmatory fingersticks required) too readily. For instance, Glucomen Day received such a status despite significant inaccuracies in its readings, particularly in the hypoglycaemic range.
- The Need for International Criteria and Standards: The IFCC team is striving to develop internationally recognised study design criteria and performance standards, leading to ISO standards akin to those for blood glucose meters. An ISO standard would allow all stakeholders to safely rely on these benchmarks to ensure CGM devices’ safety with an accepted degree of risk.
Since March 2023:
- MHRA’s Response: The MHRA reviewed clinical data from manufacturers for paediatric indications and made conclusions that demand attention.
- Medtrum A6: The MHRA has not raised significant concern over the Medtrum A6 as there is some clinical data expected to be published in 2024/2025. However, it’s notable that this device received its CE marking back in 2018. The prolonged period before publishing the clinical data suggests either an unusually long and complex trial or the possibility that this data wasn’t available when the CE mark was granted.
- GlucoMen Day: Despite concerns over the accuracy of GlucoMen Day, the MHRA has determined it’s not a significant issue due to its low usage rate, especially in individuals under 20 years old. This is controversial because general practitioners can prescribe it for children as young as six, and individuals with a group 1 license can make driving decisions based on GlucoMen Day readings above 4.0 mmol/L. Given the accuracy discrepancies presented in our analysis, this poses a question of liability if an accident occurs because of reliance on an inaccurate reading—a risk estimated to be between 10-40%, in stark contrast to the 0.2% chance with higher-performing devices.
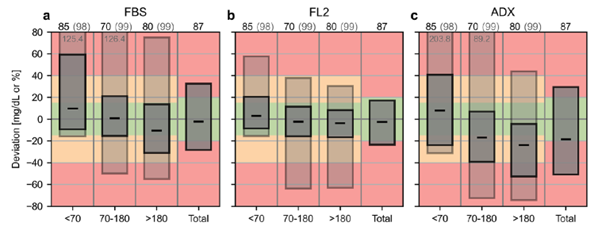
- GlucoRx Aidex: The MHRA has overlooked the study I presented, showing that the GlucoRx Aidex had a Mean Absolute Relative Difference (MARD) of over 20% and performed poorly according to the new CG-DIVA metric. By dismissing these findings and the variability of MARD, the MHRA failed to acknowledge the importance of rigorous study design and the insufficiency of assessing performance solely based on MARD.
- Diabetes Technology Meeting Insights: The IFCC presented a symposium on CGM accuracy, advocating for robust study design criteria aligned with the Dynamic Glucose Region (DGR) Plot. A consensus paper on CGM accuracy study design criteria is forthcoming that details the DGR following a published scoping review.
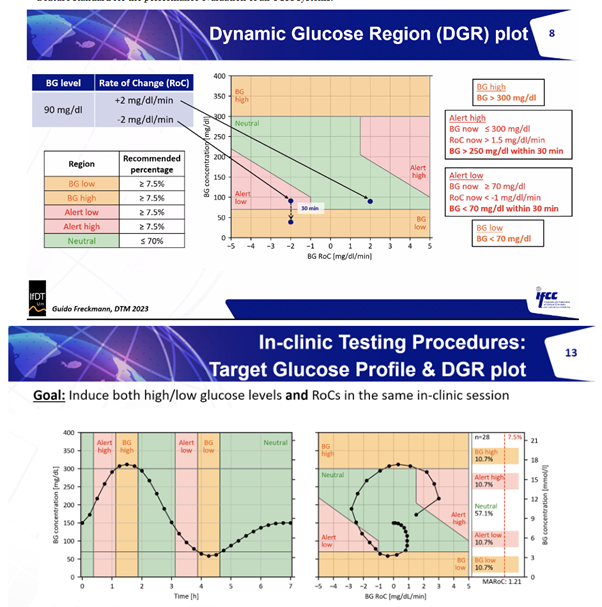
- IFCC’s CG-DIVA: The IFCC team has introduced an innovative performance metric called CG-DIVA (Great name), which transforms the criteria for integrated CGM (iCGM) systems into a visual format. To evaluate a CGM device using this metric, look for the dark boxes in the provided examples. If this box remains within the green zone, the device successfully meets the 15/15 accuracy standard. However, if the light box extends into the red zone, the device fails to achieve the more stringent 40/40 accuracy threshold. I bet you can guess which one is the FreeStyle Libre 2 and which is the GlucoRx Aidex without seeing the titles! For a more comprehensive understanding, please refer to the detailed explanation in the IFCC’s CG-DIVA publication.
- UKCA Standards Timeline: The UK seeks to establish standards by July 2024, yet CGM devices with current CE markings are exempt from updated regulations until 2028, just the same for the rest of Europe—a concerning delay.
Taking Action:
The regulatory landscape is shifting, and we must ensure the transition is swift and effective:
- Engage with Regulators: I urge you to approach the MHRA, FDA, or your local regulatory bodies, advocating for adopting the IFCC’s DGR and CG-DIVA and to support the IFCC in pushing for an ISO standard for CGM.
- Contact Key Organisations to Bolster Lobbying Power: If you have contacts at the DeDoc, ATTD, ADA, ISPAD, IDF, FDA, MHRA, DTN, JDRF (sorry for those other amazing organisations I have missed). Let them know what the IFCC is doing, and they need everyone behind them to push through an ISO standard!
- Raise Awareness: Inform and mobilise others about the urgency of implementing these standards. We can no longer tolerate devices without adequate clinical accuracy data on the market, risking the health of individuals with diabetes.
- Prepare for Advocacy: I am drafting a comprehensive letter to the MHRA detailing the imperative for adopting the DGR and CG-DIVA. Join me in this initiative—whether you’re in Europe, the United States, or elsewhere. We must let the regulators know there is a need for change, it’s time to raise consciousness.
As stakeholders in the diabetes community, we cannot be passive. It’s our responsibility to ensure that the devices our community relies on are safe, reliable, and supported by robust clinical data. This is our call to action for the health and safety of our global diabetes family.
Click to Join #WeDeserveBetter, the type 1 diabetes community (people with T1D, carers, HCPs, CGM industry) wanting a change in CGM regulation for the people, by the people!
Let’s go!
John
