The 2023 Diabetes Technology Meeting was conducted virtually from 1st to 4th November, encompassing a broad spectrum of pivotal themes in diabetes care.
The conference included a presentation I delivered on the regulatory challenges and the paucity of clinical accuracy data for Continuous Glucose Monitoring (CGM) systems in Europe and the UK. I delivered it with gusto, despite a large dose of imposter syndrome.
My Highlights from the Conference:
CGM Regulation: The ‘Wild West’ of Europe
The session initiated with my narrative review underscoring the lax regulation of CGM devices in Europe, highlighting concerns regarding the CE marking’s credibility and the absence of unified standards for study design and performance evaluation. There is an impending consensus paper on CGM accuracy study design from IFCC. The paper will detail the Dynamic Glucose Region (DGR) Plot.
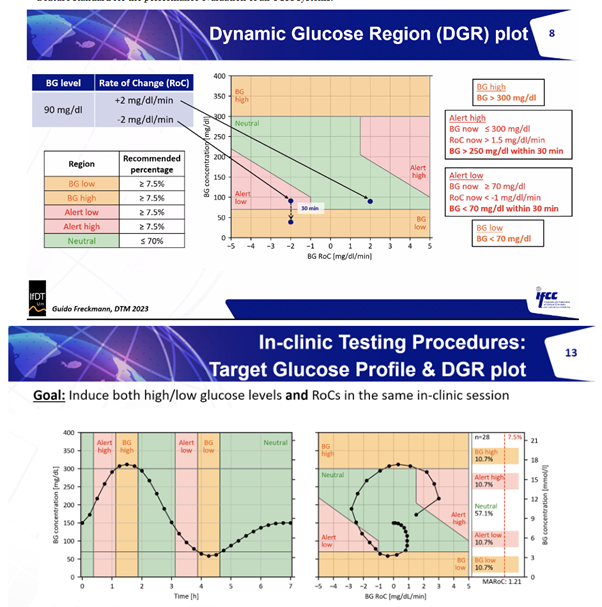
The IFCC team went on to introduce the novel CG-DIVA performance metric. Simply, “if the dark box stays in the green, it meets 15/15.” Additionally, “if the light box bleeds into the red, 40/40 is not made.”
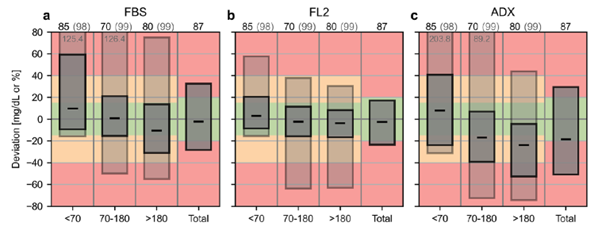
Furthermore, the session underscored the critical need for an ISO standard in this realm, with the IFCC leading advocacy efforts. A more comprehensive understanding and access to all key papers can be found here.
Are you thinking CGM regulation should not be the first highlight?
If so, consider, all future insulin decisions will be made using CGM values. Therefore, it’s essential regulation is improved to keep the risk low!
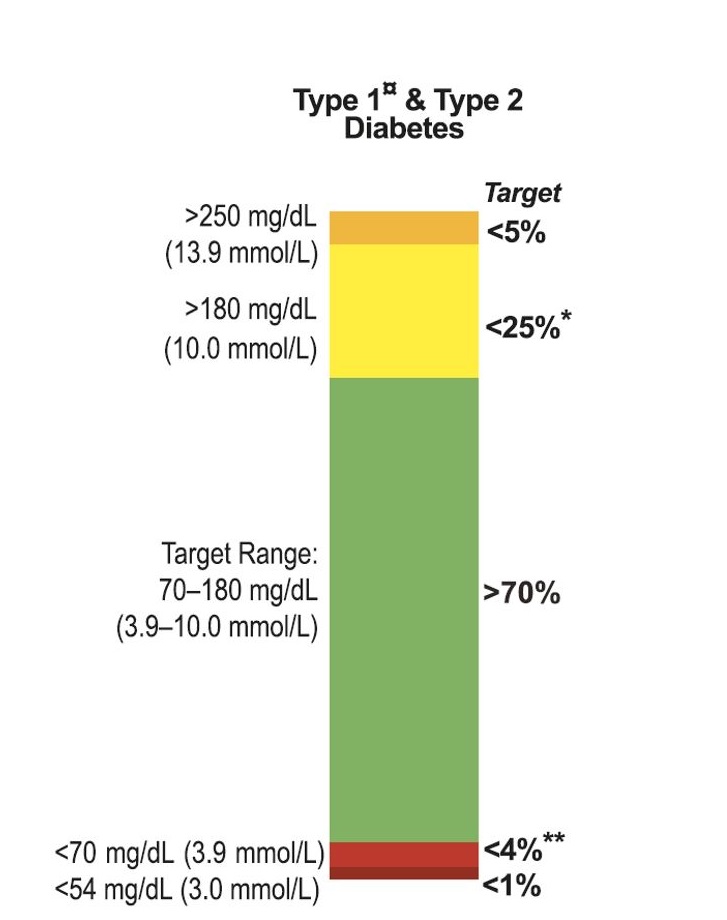
Optimising Diabetes Care: The Power of ‘Time in Range’ Metrics
Dr. Bergenstal elucidated the significance of Time in Range (TIR, 3.9-10 mmol/L or 70-180mg/dL) in managing diabetes, correlating it with a decrease in diabetes-related complications such as retinopathy. A nuanced approach to diabetes management based on TIR was proposed, with tailored strategies across four categorised zones, emphasising the management of hypoglycaemia and adjustments in therapy. There were sessions suggesting Time in Tight Range (TITR, 3.9-7.8 mmol/L of 70-140 mg/dL) will be the future! However, balanced discussion suggested the need for adding agents on top of AID systems before this happens ubiquitously (more to come on dual therapy).
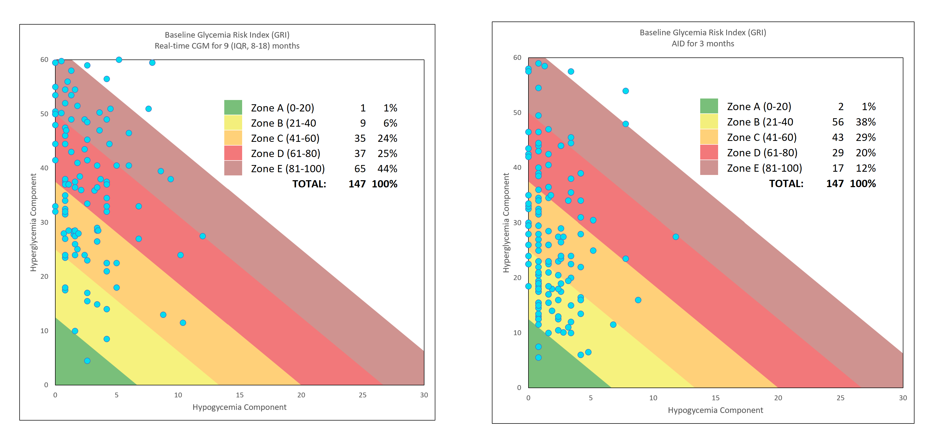
Glycemic Risk Index: A Novel Benchmark for Control Assessment
Dr. David Klonoff introduced the Glycemic Risk Index (GRI), a tool for evaluating the quality of glycemic control, with implications for both clinical practice and research. This chart shows the GRI’s utility in assessing children transitioning from CGM to Automated Insulin Delivery (AID) Systems.
Enter your CGM metrics to generate a Glycemic Risk Index score and zone. Then make changes to level level up (well down to be technically correct!).
CGM Benefits those not living with diabetes
Dr. Tracey McLaughlin’s highlighted the advantages of CGM use in individuals without diabetes. The study presented showcased improvements in blood glucose stability and reductions in extreme glycaemic events. Furthermore, weight loss benefits were observed, indicating the broader utility of CGM in preventative healthcare. Not to mention, the market is worth billions! Watch this space!
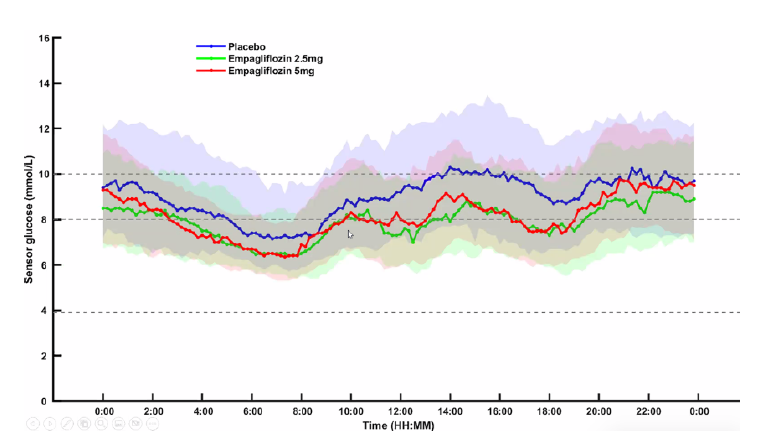
Dual-Agent Therapy in Type 1 Diabetes Enhances TIR
Dr. Haidar presented a study on the efficacy of combining SGLT-2 inhibitors with Automated Insulin Delivery (AID) in Type 1 diabetes management. The study reported significant improvements in TIR and plans for future research into the combination of semaglutide with AID. Dual therapy might change the go to metric from TIR to TITR?
Technological Advancements in CGM Devices
The conference also featured updates on the latest CGM technologies. Abbott’s FreeStyle Libre 2 Plus sensors gained FDA approval, boasting high accuracy and extended wear time. Dexcom’s partnerships underscored the potential for CGM in broader metabolic health contexts, while Medtronic’s Guardian 4 Sensor was lauded for improved user experience. Senseonics introduced new products with enhanced capabilities, and Pacific Diabetes Technologies presented the innovative SynerG device, a combination CGM and insulin pump, currently under further investigation.
The Diabetes Technology Meeting 2023 was a comprehensive convergence of experts discussing advancements, regulatory updates, and future directions in diabetes technology. The emphasis on establishing international standards and supporting initiatives to enhance the safety and efficacy of diabetes technology was a recurring theme.
Cheers
John
