Insulin Resistance Guide — Part 3
Seven Ways to Improve Insulin Sensitivity
The seven practical levers that most reliably shift insulin resistance in type 1 diabetes — lifestyle foundations first, then pharmacology where appropriate.
Overview
Insulin resistance in T1D is rarely one thing. It usually emerges from overlapping disturbances across muscle, liver, adipose tissue, gut hormone signalling, and brain insulin sensitivity. This page focuses on what tends to shift the system meaningfully in the real world.
The seven pillars below are stacked like a pyramid: lifestyle foundations first, then pharmacology where appropriate.
Prefer audio? Listen to GNL Podcast Episode 14: Overcoming Insulin Resistance.
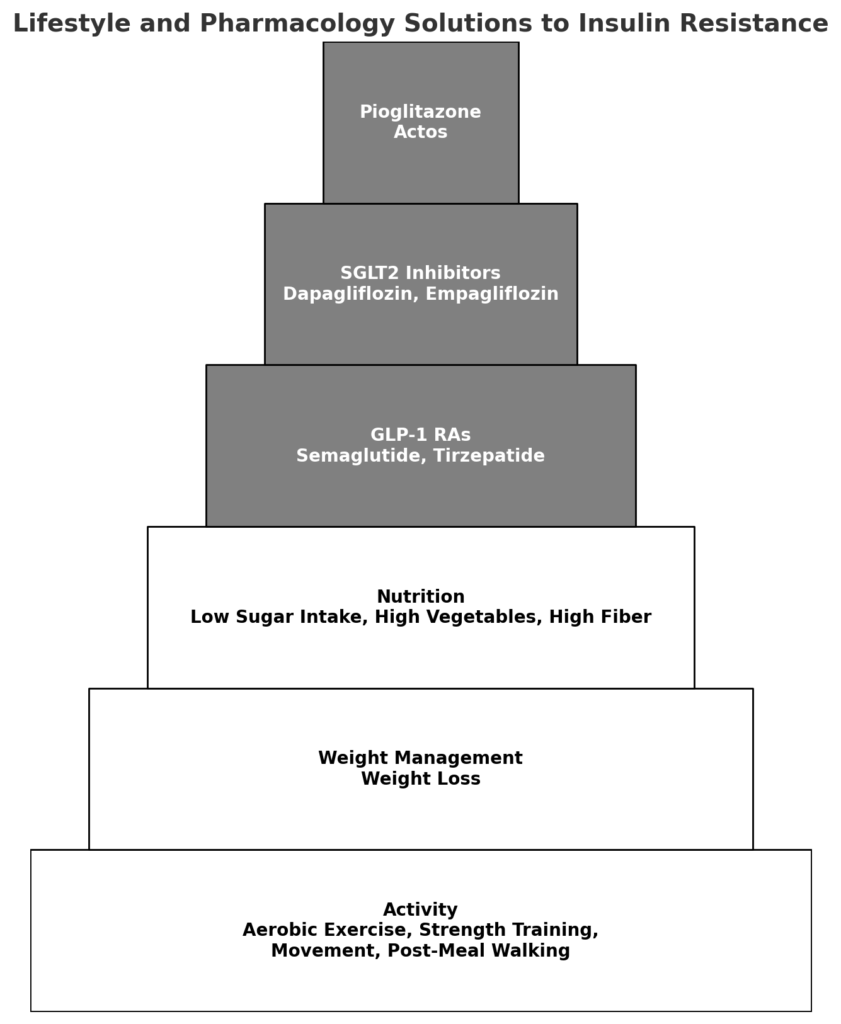
The organising principle
On average, insulin resistance tends to improve most through (1) movement, (2) body fat reduction where appropriate, and (3) reducing glucotoxicity. Pharmacology can be powerful, but it typically works best when layered onto these foundations.
The art is choosing the smallest set of levers that meaningfully shifts the trajectory. Below are all seven pillars.
Pillar 1: Activity (aerobic + resistance + daily movement)
Physical activity improves insulin sensitivity through several mechanisms at once: increased glucose uptake (including non-insulin-mediated uptake), improved muscle insulin signalling, reduced intramuscular lipid burden (DAGs), improved hepatic insulin sensitivity, and improved appetite regulation via brain signalling.

- Fasted exercise: Morning exercise performed at low circulating insulin levels may favour greater fat oxidation and often improves whole-day insulin sensitivity.
- 10-15 minutes after meals: This tends to reduce post-meal peaks and supercharge the circulating meal insulin, so less is required for the same effect.
- Strength training 3-4 times per week: Increases muscle mass and glucose disposal capacity — this is where many people find the most leverage.
- Aerobic training 3-5 times per week: Helps clear intramuscular lipid burden (DAGs) and improves hepatic control.
- Daily movement: Breaks sedentary time and keeps insulin action more predictable.
- Activity snacking: Small bursts of movement that reduce glucose and metabolic load without requiring a full workout.
Important: increasing activity often requires pre-planned insulin reductions. Without this, the intervention can become a hypoglycaemia generator rather than a sensitivity improver.
Resources:
Pillar 2: Weight management (fat loss where appropriate)
Where excess body fat (especially visceral and ectopic fat) is present, fat loss can materially improve insulin sensitivity. Even 5-10% weight loss tends to be meaningful; 15-20% can be transformative for hepatic and muscle insulin resistance in many people.
The goal is fat loss with muscle preservation.
- Protein: Many people aim for around 1.5-2.0 g/kg/day to support satiety and preserve lean mass.
- Resistance training: At least 3-4 times per week while losing weight.
- Energy deficit: Often around 500-1,000 kcal/day for approximately 0.5-1.0 kg/week loss — this varies between individuals.
- Sleep and stress: Poor sleep and chronic stress tend to make fat loss harder and can worsen insulin resistance independently.
Pillar 3: Nutrition (reduce glucotoxicity and lipotoxicity)
Nutrition influences insulin resistance through energy balance, glucose exposure (glucotoxicity), lipid exposure (lipotoxicity), and gut hormone signalling.
- Limit liquid sugar and fructose-heavy inputs — this is a common lever on hepatic de novo lipogenesis. For a deep dive, see Attia #87 with Rick Johnson on fructose.
- Prioritise fibre-rich whole foods to slow absorption and improve satiety.
- Watch for saturated fat load where delayed spikes and insulin resistance patterns are prominent.
- CGM feedback loop: CGM patterns can help refine meals over time — not to chase perfection, but to spot patterns. See the CGM accuracy guide.
- Pre-bolus timing: Often around 15-20 minutes before meals tends to improve matching, though this varies widely with individual factors like gastroparesis and AID use. See the Bolus insulin guide.
- Hypo treatment: Glucose-only options tend to be more predictable than mixed sugars that can stack unpredictably.

Pillar 4: GLP-1 receptor agonists and dual agonists
GLP-1-based therapies (such as semaglutide and tirzepatide) are a legitimate pillar for insulin resistance management when obesity and/or appetite dysregulation are part of the picture. Most use in T1D is currently off-label in many systems.
Beyond weight loss, the mechanistic reasons these may help in T1D include: reduced glucagon output, reduced hepatic glucose output, improved satiety signalling, and slower gastric emptying (which can improve matching when handled carefully).
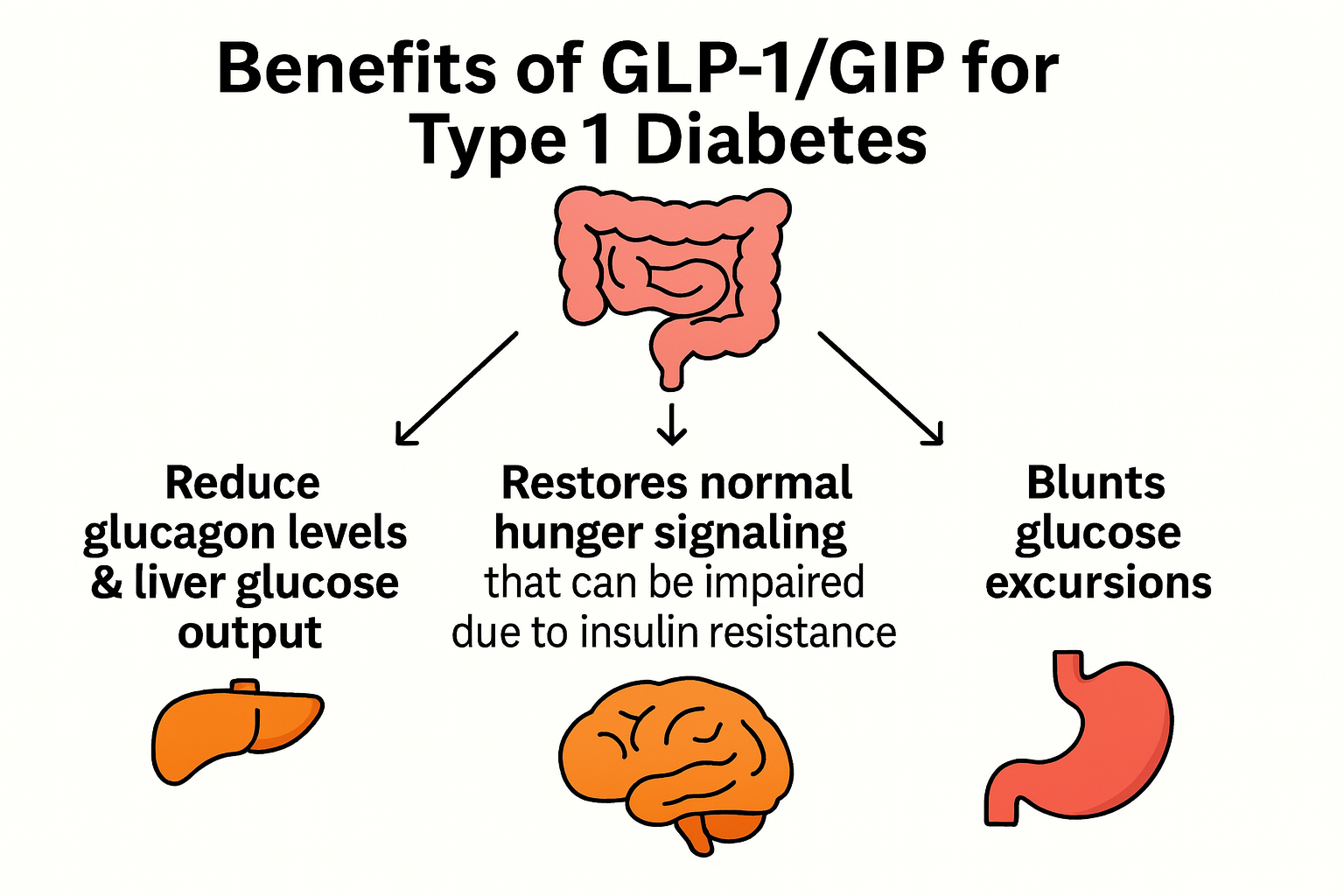
Safety considerations specific to T1D
- Insulin requirements can fall quickly — reductions of around 30% are commonly discussed in practice. Without planned reductions, hypoglycaemia is likely.
- Slower gastric emptying can make bolus timing harder, especially off AID systems.
- Muscle preservation matters during weight loss: pairing with protein and resistance training is important.
This is firmly in the territory of working closely with a clinician and following a structured protocol.
Resources:
Pillar 5: SGLT-2 inhibitors
SGLT-2 inhibitors lower glucose by increasing urinary glucose excretion, reducing glucose toxicity, and often reducing insulin requirements — without directly increasing insulin.
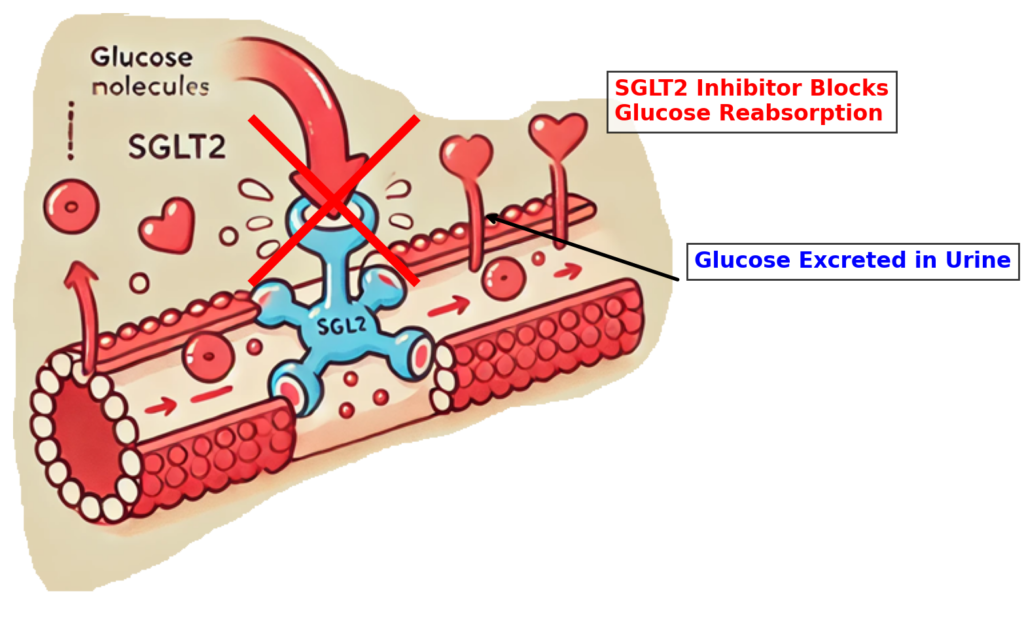
- Potential benefits: Lower glucose exposure, less insulin required, sometimes improved variability.
- Major risk in T1D: DKA (including euglycaemic DKA) remains the primary concern when used without strict education and protocols.
- Practical requirements: Hydration, ketone monitoring, sick-day rules, and clear stop rules are non-negotiable.
This is firmly territory to discuss with your diabetes care team — the DKA risk is the price of entry.
Pillar 6: Pioglitazone (PPAR-gamma agonist)
Pioglitazone can improve insulin sensitivity partly by redistributing fat away from ectopic depots (liver and muscle) towards safer subcutaneous stores. It is often avoided because of weight gain concerns, but the mechanism is more nuanced than that label suggests.
- Potential upside: Improved hepatic and muscle insulin signalling.
- Trade-offs: Fluid retention risk (and other contraindications), slower onset (weeks to months), and careful patient selection.
- Best paired with resistance training and adequate protein to protect lean mass.
Pillar 7: Metformin (mostly hepatic)
Metformin is often described as a general insulin sensitiser, but mechanistically, its clinically relevant effects are largely hepatic (liver). It reduces hepatic glucose output and can reduce glucose toxicity. It does not directly address muscle insulin resistance.
- The typical insulin-lowering effect in T1D is often modest — commonly cited around 5% in practice summaries.
- Still useful in a multi-lever approach because it is inexpensive and has a long safety record (with appropriate monitoring and contraindications respected).
Mechanism deep dive: Attia #337 with Ralph DeFronzo — insulin resistance masterclass
What this means in practice
For most people, the fastest return comes from the lifestyle foundations: (1) movement, (2) fat loss where appropriate, and (3) reducing glucotoxicity. Pharmacology tends to work best when layered onto these foundations rather than used in isolation.
- When increasing activity: Many people find that pre-planned insulin reductions prevent hypoglycaemia from becoming the limiting factor.
- Nutrition shifts: Favouring fibre-rich foods, limiting saturated fat load, and maintaining a modest energy deficit (around 500-1,000 kcal/day) can meaningfully reduce lipid-driven insulin resistance over time.
- When pursuing fat loss: Protecting muscle through adequate protein (around 1.5-2.0 g/kg/day) and resistance training (3-4 times per week) tends to preserve the metabolic benefit.
- GLP-1 and dual agonists: Insulin needs can fall quickly (often around 30% early on), and bolus timing may need adjustment due to slower gastric emptying.
- SGLT-2 inhibitors: Ketone education, sick-day rules, hydration, and clear stop rules are the non-negotiable foundation for safe use.
The goal is not to do everything. The goal is to choose the smallest set of levers that measurably reduces insulin dose requirements, improves predictability, and lowers glucose exposure over time — worth exploring with your diabetes care team.
This content is for educational exploration only. It describes average responses and general principles. It is not medical advice and cannot replace individual clinical guidance from your diabetes care team.

